Pink diamonds from the Argyle mine have been known by polishers, laboratories, and traders to exhibit a phenomenon of photochromism whereby their color can be influenced by the illumination of different lights and intensities. These influences are relatively short-lived and most conspicuous after illumination by short wavelength light of high intensity. This effect is particularly noticeable when using instruments to observe fluorescence, such as a DiamondView, which results in a distinctively bleached or pale colouration when used with Argyle pink diamonds.
Color intensification can be achieved by subjecting a pink diamond to elevated temperature, called thermo-chromism. A diamond’s color can also change unpredictably during polishing, a phenomenon known as tribo-chromism. All of these possible changes are temporary, and the diamond’s color can readily return to an ‘ambient resting color’.
References to pink diamonds in this article apply primarily to Argyle pink diamonds (Figure 1) rather than Golconda-style Type II pinks or Type I purplish diamonds from Siberia or Canada.
Figure 1: An intensely saturated pink diamond from the Argyle mine in Western Australia. Photo courtesy Rio Tinto Diamonds.
Photo-chromism
While anecdotal reports have been made about color changes in Argyle pink diamonds, the results of studies by Noble, Byrne, and Fisher (Noble 2011, Byrne 2012, Fisher 2009) have provided a much better understanding of the phenomenon, in which electrons are transferred between defects, one of which is responsible for the pink coloration. Bleaching and restoration of color are the better-characterized phenomena relating to pink color changes. Generally speaking, UV illumination tends to bleach a pink diamond’s color, whereas visible light restores color. The shorter the UV wavelength (down to nearly 300 nm), the greater the extent of the bleaching, whereas the intensity of the bleaching or restoration light affects the rate of color change. Even prolonged periods of time of days or longer in darkness have not been found to result in any recovery of color.
Raising a pink diamond’s temperature to above about 800˚C intensifies its color and makes it appear more purplish. As seen with bleached diamonds, exposure to white light restores the color. Blue light has been found to be most effective at restoring color, particularly near 460 nm. The images in Figure 2. show the extent of color changes that can be achieved.
Figure 2: The color of pink diamonds can be deepened (left) or lightened (right) and returned to their original ‘daylight’ color (center) after illumination with white or blue light. The brown diamonds are for color reference.
Figure 3: A pink diamond becomes bleached during polishing, though the effect is short-lived and recovery is unaffected by darkness.
Tribo-chromism
During polishing, a diamond can experience temperatures that result in the gem glowing, depending on the force applied to the gem on a polishing wheel and the crystallographic orientation of the face being polished. However, color changes are commonly observable immediately after polishing (Figure 3). These tribo-chromism changes include very short-lived bleaching that lasts only for a few seconds. Strangely, this change is followed by a longer-lived deepening of the color. This may be the reason that several polishers have claimed that laboratories somehow altered the color of the pinks they submitted for grading.
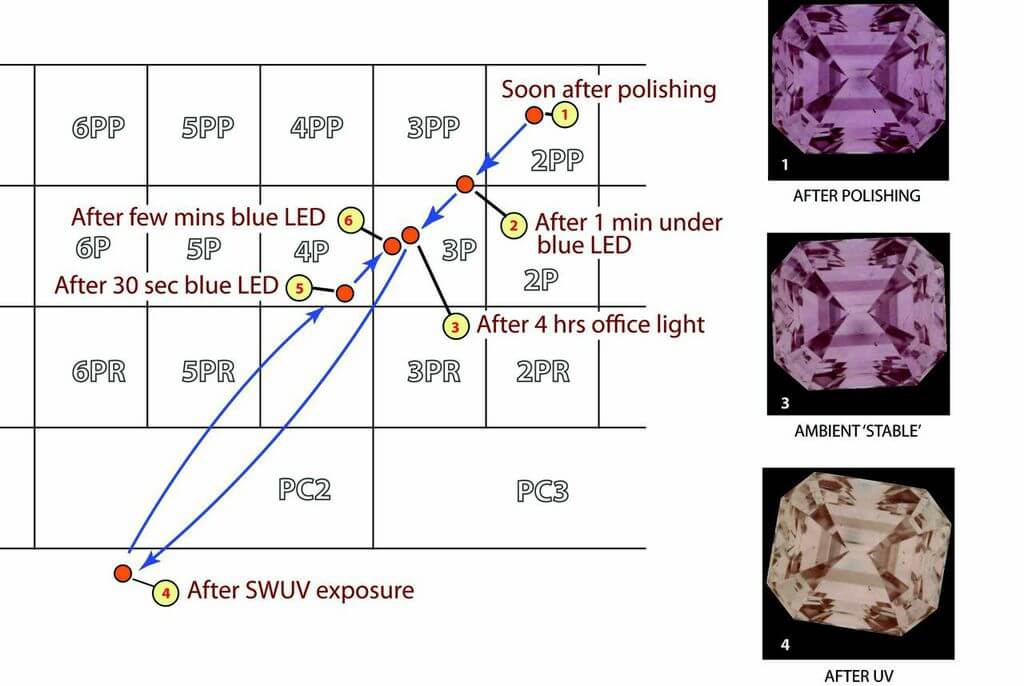
To illustrate the effect of color change after polishing, exposure to UV light and to blue light, a pink diamond fresh off a polishing wheel was presented to a (proprietary) objective color grading system, after which it was illuminated with blue light, and again presented to the color grader. The stone was then illuminated with SWUV and finally, blue light with color grade measurements made at each stage. Figure 4. shows how the color varied from the 2PP to ‘champagne’ (light brown). The slight disparity between stages 3 and 6 reflects either instrumental error or insufficient exposure to blue light at stage 6.
Figure 4: The movement of an Argyle pink across the Argyle color palette in a sequence that comprises polishing, bleaching with SWUV, and exposure to blue light.
Significantly, preconditioning with blue light provided stability for grading the color, irrespective of whether the color was deepened or bleached.
Color Grading
At wavelengths below ~ 460 nm can influence color, the spectrum of a grading light can induce subtle changes to a stone’s color and hence its grade, depending on the reference and method used for grading.
Color grading of diamonds is normally undertaken under a controlled lighting condition and with reference to either a master set of stones or ‘color chip’ such as a Munsell Color Card. From the previous discussion, it is clear that the spectral characteristics of the lighting, especially in the blue and UV regions, can affect the color of the gem being graded and/ or of a master set if assembled from Argyle-type pinks. If both the graded gem and the master set are exposed equally under the grading light, then color changes are not critical, as matching stones will be equally affected. However if either a master set or a pink diamond is subjected to a grading light for a prolonged period of time, the resulting color changes may cause the subject stone to cross a grading border.
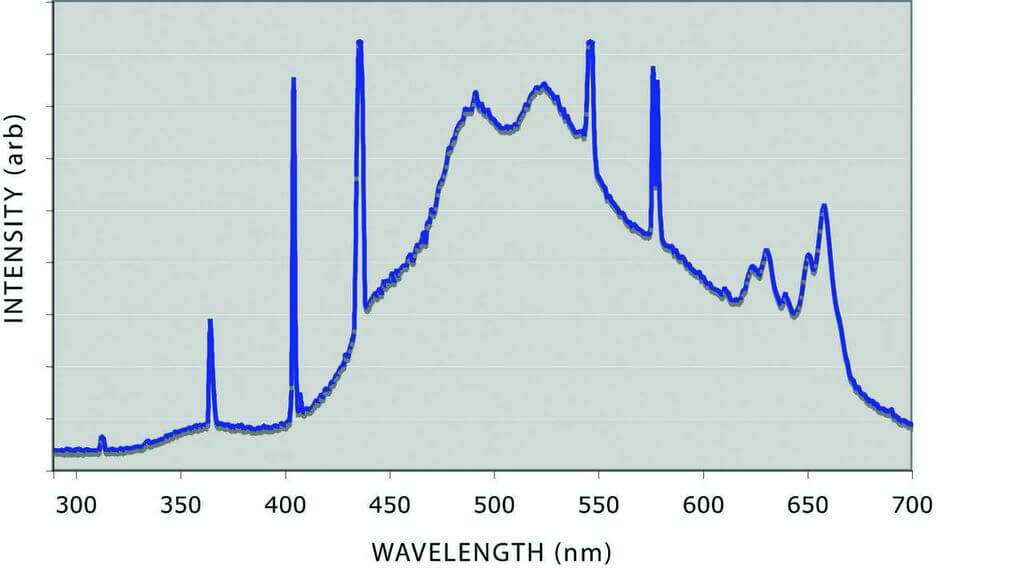
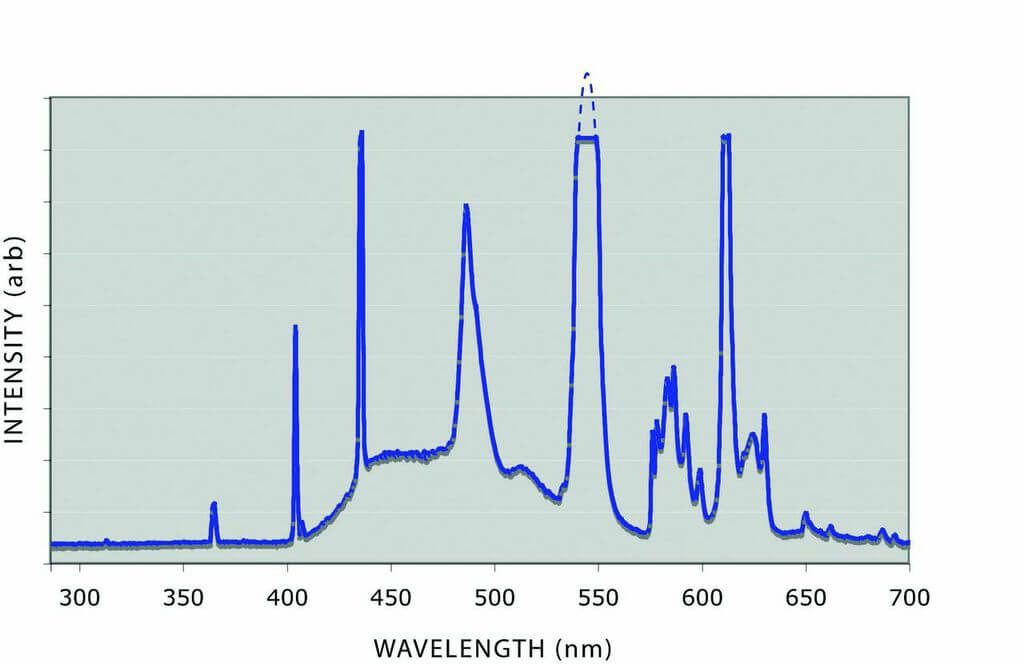
The spectra from two commonly used grading lights are shown in Figure 5. Both are rated as having a daylight temperature of 6500K. One of the sources shows a much higher UV component than the other. A test with a 2PP pink placed in the lighting environment of each of the lamps resulted in a change of measured color by one-quarter of an Argyle color grade, sufficient to cross a color boundary.
Figure 5: The spectra of two 6500K daylight fluorescent tubes showing different levels of short wavelength light
Daylight outside shows a much larger UV component than the grading lamps, however window glass is very effective at absorbing wavelengths below 400 nm, so wearers of pink diamonds may notice an improvement in color when they go inside from the outdoors!
If a pink diamond is left in the dark, its color will not change regardless of its color state (except in instances of a diamond recovering from tribo-chromic bleaching). Curiously, the color of a pink stone that was bleached cannot be restored by subjecting it to elevated temperature alone. Restoration with visible light is required in these instances. The processes taking place at the electronic level are not sufficiently understood to explain these phenomena, and studies of them may shed further insight into the structure of the color center responsible for pink diamonds, the details of which still remain a mystery.
Conclusion
The extremely high value placed on Argyle pink diamonds demands a high level of understanding of the factors that influence color, especially during color grading. The color of pink diamonds from the Argyle mine and other deposits is affected by the balance of bleaching wavelengths below about 450 nm, and by color restoration wavelengths above 450 nm. Color is particularly prone to bleaching from UV wavelengths. Additionally, the initial color state of diamonds can be darkened by the polishing process. Consequently, caution must be taken when color grading a pink diamond based on its prior history and the lighting conditions. Laboratories must be vigilant in the case of freshly-polished stones, and stones being examined for fluorescence.
Restoration of a diamond’s pink color to an ‘ambient daylight’ state can be achieved by exposure to white light or accelerated by illumination from a high intensity blue LED with a wavelength near 460 nm.
References
Byrne K.S. et. al., 2012. Optically Reversible Photochromism in Natural Pink Diamond. Diamond and Related Materials Vol.11. 2012 pp.30, 31-36.
Noble C. J. 2011. Applications of Magnetic Resonance in Materials Science and Solid State Physics. PhD thesis, Monash University.
Fisher D et al., 2009. Brown Color in Natural Diamond and Interaction between the Brown Related and other Color-inducing Defects. J. Phys: Condensed Matter, 21:364213, 2009.
The Argyle Pink Diamond grading system
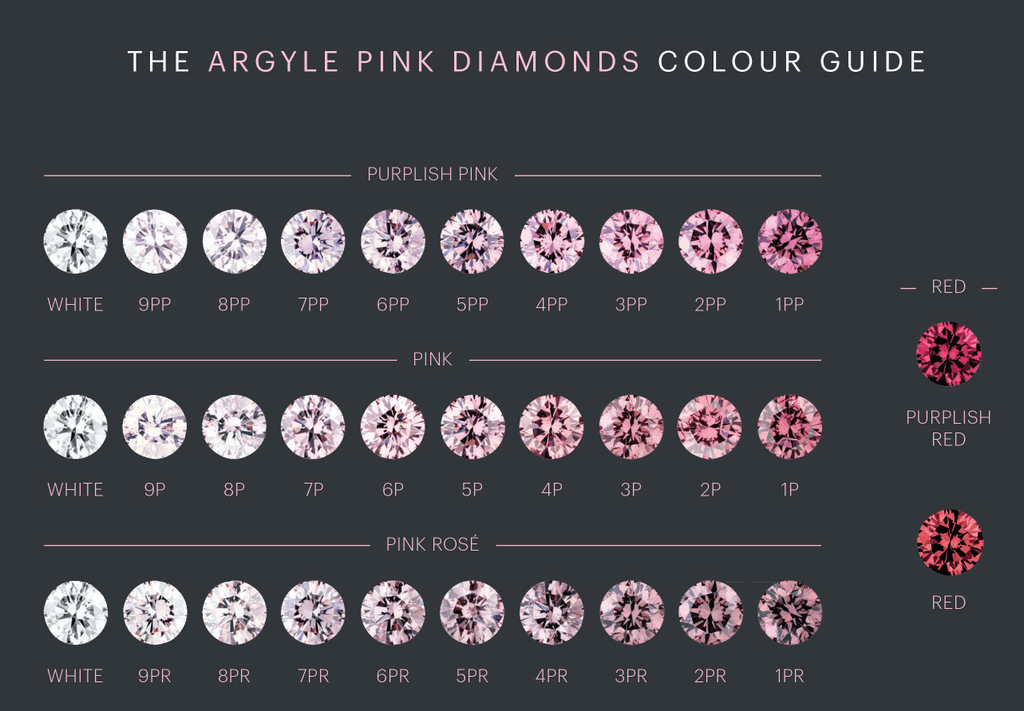
The prominence of Argyle Diamonds in marketing pink diamonds led to the development of a color grading scale particularly suited to the product. It assigns diamonds a color grade which refers to hue and saturation. Three hues are recognized comprising purplish-pink (PP), pink (P), and pink-rose (PR), the latter having a brownish secondary color. A numerical prefix is added to the hues representing the degree of saturation, where 1 is the highest and 9 the palest. For saturations higher than 1, the grade may transcend into red or purplish red.
This article originally appeared in The Australian Gemmologist (Fourth Quarter of 2014), volume 25, 8. All images courtesy of Rio Tinto Diamonds.